 
Cows need sodium bicarbonate to properly digest fiber and sustain the pH of the rumen. For poultry, this valuable supplement enables them to tolerate heat, increases the quality of the eggshell, and keeps up the balance of sodium. Much of sodium bicarbonate production goes toward feed supplements for animals, accounting for approximately 25% of its use. Carbonated soda manufacturers have taken advantage of this reaction and frequently add baking soda to drinks to create carbon dioxide bubbles. 
This makes it the most commonly used leavening agent in cooking. When used in baking as a key ingredient in baking powder, sodium bicarbonate reacts and releases carbon dioxide. In the United States alone, 33% of baking soda production is consumed in this manner. The reaction of baking soda and vinegar is a favorite science fair project to make a volcano eruption.Īlthough many people associate baking soda with its role as a cleaner and deodorizer, its chief use is in the food industry. Nahcolite only forms when there is excessive and contained carbon dioxide otherwise, trona will form in its place. When water vapor and carbon dioxide react with trona, sodium bicarbonate is a resulting byproduct. Sodium bicarbonate is rarely found in nature because, when exposed, it loses carbon dioxide and passes into another mineral called trona (hydrated sodium bicarbonate carbonate). In the oil-shale riverbed of the Green River (Colorado), nahcolite deposits as large as five feet in diameter and other formations with layers four inches thick, have been discovered. Although the first major find of nahcolite was near Searles Lake, California in 1940, it was first reported by P. Nahcolite has been found most notably in California, Colorado, Egypt and Kenya. 
Nahcolite coloring ranges from colorless to brown, and the crystals are monoclinic and transparent. This mineral, known as an evaporite (“A sedimentary deposit that results from the evaporation of seawater”), forms in oil shales and lake sediment deposits, and capable of absorbing water. The name Nahcolite is a mnemonic for its chemical formula (Na+H+CO+lite). 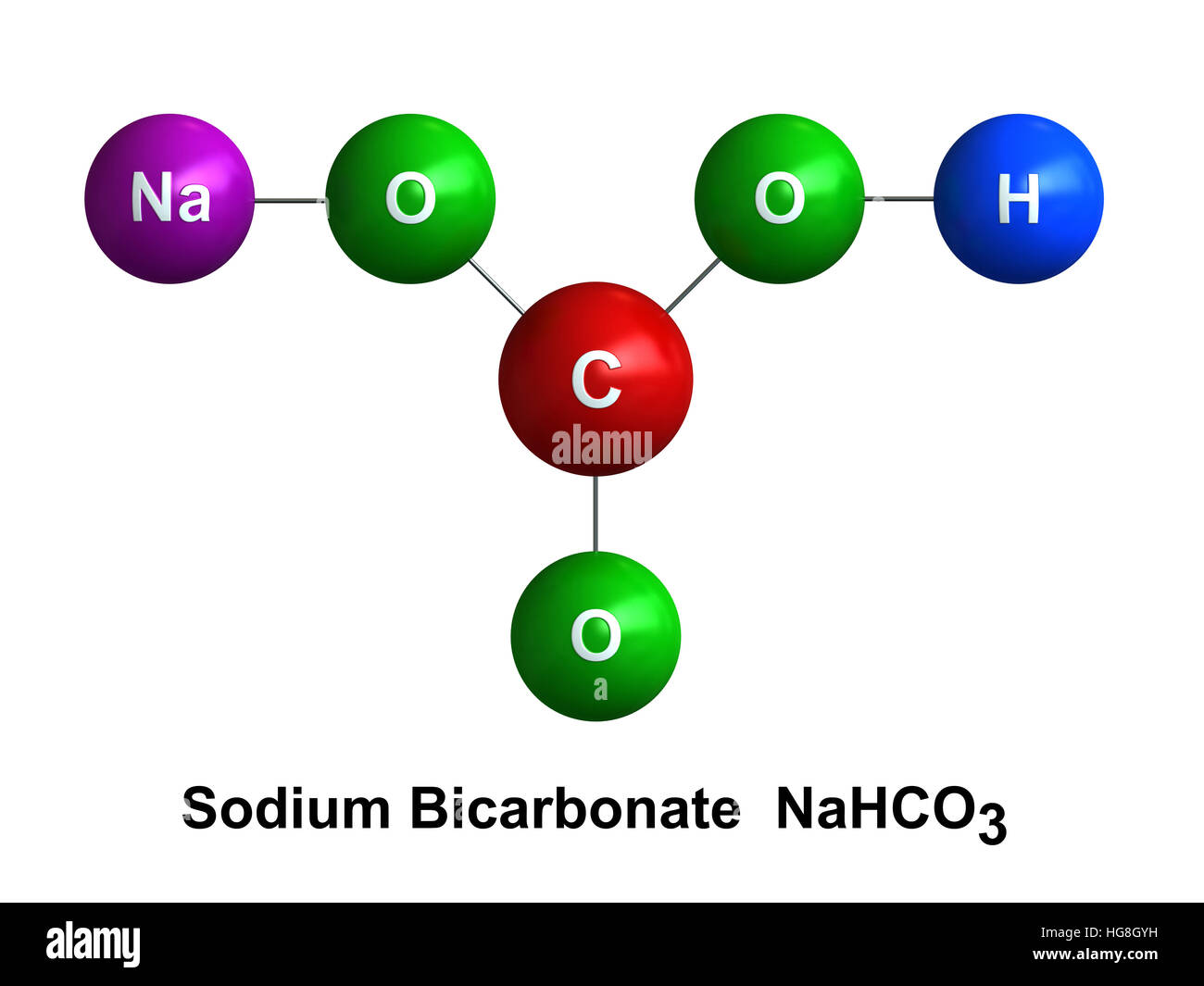
Most sodium bicarbonate is produced through artificial means, but in nature it's found in a mineral called nahcolite. Although it is a mild irritant to the eyes, baking soda remains otherwise non-toxic and safe for general use. Its chemical attributes also prevent oxidization and make it insoluble (doesn't dissolve) in alcohol. Baking soda is neither flammable nor explosive. Its decomposition produces sodium carbonate (Na 2CO 3), carbon dioxide (CO 2), and water (H 2O). Because sodium bicarbonate begins to decompose at 50☌, it is difficult to properly determine a boiling or melting point. Baking soda exists as a solid composed of white, monoclinic crystals and weighs 84.01 g/mol. Sodium bicarbonate is capable of precipitating calcium and can absorb acid gas emissions such as sulfur dioxide. Carbon dioxide is heavier than the air fueling the fire, so the baking soda smothers the flames. When heated, sodium bicarbonate releases carbon dioxide and it reacts as a leavening agent for culinary purposes as well as a fire retardant. This classification results from the combination of the base sodium hydroxide and the acid carbonic, which also gives baking soda the properties of a mild alkali. Sodium bicarbonate (most commonly known as baking soda) is classified as an acid salt. This natural substance has also increased in popularity because it has no adverse effects on the environment. Baking soda was first used mainly for cooking purposes, but over the last 150 years, its production has grown to include manufacturing of other metals and glass, and as a basic household cleaner. Found in deposits throughout the globe, natural sodium bicarbonate works to maintain the Earth's balance of carbon dioxide and pH. Sodium bicarbonate (NaHCO 3), recognized by most as ordinary baking soda, is a compound known for its unusual properties and diverse applications. Materials in their standard state (at 25 ☌, 100 kPa) Except where noted otherwise, data are given for 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
